Some of the most brilliant scientists and forward-thinking people on the planet see a new way forward: bringing back extinct animals. Learn the scientific and ethical reasons bioengineering a new wave of species will help the world heal.
Inside This Article:
- Why CRISPR is the linchpin of de-extinction
- 12 steps for resurrecting extinct species
- How the Woolly Mammoth will heal the planet
What Is De-Extinction?
De-extinction, or resurrection biology, reverses plant and animal extinctions by creating new versions of previously lost species. Back-breeding, cloning, and genome editing are species restoration methods. The goal is to re-establish dynamic processes that produce healthy ecosystems and restore biodiversity.
In 2016, The International Union for the Conservation of Nature (IUCN) drafted the IUCN SSC Guiding Principles on Creating Proxies of Extinct Species for Conservation Benefit. In addition to providing a framework for scientists and other stakeholders, the guidelines explain that the term de-extinction is somewhat misleading. It implies that it is possible to resurrect extinct species in their genetic, behavioral, and physiological entirety. This is not the case.
National Geographic Society hosted the first TEDxDeExtinction event in March 2013, bringing public attention to de-extinction technologies. Since then, efforts like the Woolly Mammoth and Passenger Pigeon de-extinction projects have prompted public and scientific interest. A wealth of scientific literature has sparked a vigorous debate on the ethics of extinct species restoration.
How Does De-Extinction Help the Ecosystem?
Conservationists frame de-extinction as ‘deep ecological enrichment’ or restoring ecosystem functions lost through extinction. This idea is the catalyst for de-extinction projects: releasing resurrected animals into suitable habitats increases biodiversity and ecosystem resilience.
Colossal’s disruptive conservation project aims to introduce mammoth genes into the Asian Elephant for conservation purposes as a prime example. “De-extinction through genetic engineering is an incredible milestone,” asserts George Church, Ph.D., Harvard and MIT geneticist and Founder of Colossal. “But what’s more important is the impact it has on restoring habitats for carbon absorption and sequestering, like the Woolly Mammoth and Arctic grasslands.”
The return of the Woolly Mammoth to the Arctic tundra it roamed thousands of years ago has significant benefits for combating climate change. Mammoths will stir up the icy surface of the landscape, stomp out-thick, low-oxygen trees, and expose healthy, carbon-trapping grasses. That action can restore the tundra, which in turn protects the climate and balances greenhouse gasses.
How Is De-Extinction Possible?
De-extinction is possible because of bioengineering techniques that can manipulate DNA and genomes or the genetic material in an organism. Recovered DNA, whether it is from the Pleistocene or the twentieth century, makes de-extinction a reality.
“Traditional conservation efforts, while vital, have been slowly losing ground,” notes Colossal’s Church. “Species de-extinction and preservation through gene-editing technology represents an exciting and tangible new movement in science and conservation. It’s one we hope catches the public’s eye and opens doors to a new generation of molecularly-inspired conservation capable of gaining ground and reversing loss.”
What Is the Science Behind De-Extinction?
The biotechnology in place to create mammal proxies include back-breeding, cloning, and gene editing. Genetic engineering, specifically CRISPR, is producing positive results in addressing de-extinction.
Ways de-extinction science work:
- Back-breeding: Back breeding or breeding back, or selective breeding, uses the principles of selective breeding. For centuries, scientists have been using this process to resurrect diluted or lost animal traits. In 1920, German zoologists Heinz and Lutz Heck tried to breed an aurochs facsimile, a European wild ox, an ancestor of modern-day cattle. Selective breeding can increase the presence of specific traits within a population. Back-breeding has limitations as a de-extinction approach. For example, back-breeding picks out ancestral characteristics found in a living species. This method works well when the extinct species are closely related to a still-living species. In addition, there is no guarantee the selected characteristics will occur since the current environment is likely far different from when the extinct species walked the earth. For instance with back breeding cattle to help serve the goal of natural grazing, we may get cattle that resemble the auroch, but we will never get cattle that are the auroch genetically. They will remain two separate species. Back-breeding may have the ramification of a higher degree of inbreeding or gene mutation; both may reduce the population’s overall health and viability.
- Cloning: Breakthroughs in cloning, or processes that produce genetically identical biological life copies, have grown over time. In 1902, Hans Adolf Eduard Driesch, a German scientist, gave life to salamander twins by dividing an embryo into two separate viable embryos. After decades of experimentation, on July 5, 1996, a lamb named Dolly was born from a female sheep—the first mammal cloned from adult animal cells. The scientists used the somatic cell nuclear transfer (SCNT) technique to generate an identical nuclear gene sequence to the donor of the somatic (non-reproductive) cell. Cloning is a feasible de-extinction approach for living species close to extinction because the resulting organism is identical. Cloning requires intact living cells; the process is more suitable for populations where some individuals remain, not those already extinct.
- Genome Editing and Synthetic Genomics: Genetic engineering has been in development since the 1970s. Genome editing, first developed in the 1990s, manipulates a living organism’s genetic material by deleting, replacing, or inserting a DNA sequence. Scientists use tools to make small changes to the organism’s DNA. Genome editing tools delete or add small sections of DNA in the genome, the complete set of genes in an organism. DNA manipulation leads to changes in physical traits, like eye color and disease risk. Synthetic genomics is similar to genome editing because both change genetic coding. However, scientists usually attach long sections of DNA and introduce them into a biological entity’s genome. These synthesized DNA pieces can be novel genes or genes found in other organisms.
How De-Extinction Works through Genome Editing
Genome editing technology creates hybrids between living and extinct organisms. Scientists insert edited DNA from an extinct species into the nucleus of a reproducing cell. They use this technique to resurrect more species, including those whose remains are not well-preserved.
Genome editing blends the desired traits that made the species unique with genes from the donor species. That is why the resulting organism is not completely identical to the extinct species but is a hybrid.
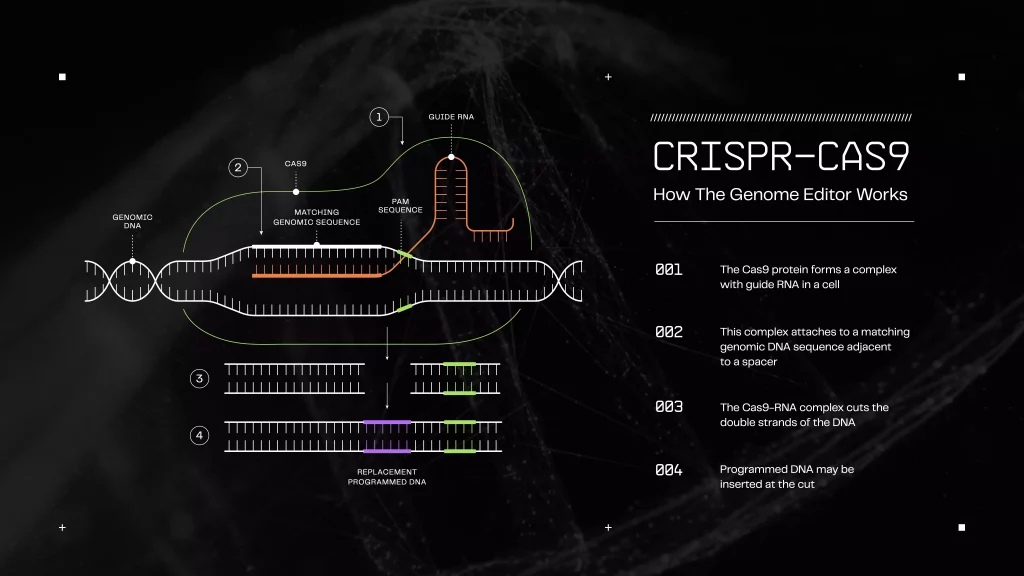
CRISPR Genome Editing and De-extinction
Clustered regularly interspaced short palindromic repeats (CRISPR) is an engineered cellular technology. CRISPR has two main functions: an RNA guide or libraries that scientists program to target specific locations on a genome and the Cas9 protein that acts as molecular scissors. In a way, CRISPR is a cut-and-paste tool that deletes or adds genetic information. The tool recognizes and cuts specific DNA inside a cell nucleus. The cuts activate repairs so that scientists can edit DNA.
We’ve found a way to harness CRISPR’s power for species de-extinction,” says Colossal’s Church. “However, gene editing has the potential to impact all aspects of life–from animal de-extinction and ecosystem restoration, to disease prevention and creating more sustainable human bodies.” Colossal has a Woolly Mammoth project for de-extinction and climate change mitigation well underway.
Step-by-Step Woolly Mammoth De-extinction Process
The Woolly Mammoth de-extinction project leading the charge to the world’s first feasible species restoration approach consists of 11 steps. The process ends in the birth of a Woolly Mammoth, which Colossal aims to achieve within four to six years.
Making Woolly Mammoths
Dr. Church answers two questions: “How will de-extinction work to bring back mammoths?” and “How will mammoths help mitigate climate change?”
In How to Clone a Mammoth: The Science of De-extinction, Beth Shapiro, Ph.D., evolutionary biologist and ancient DNA specialist, confirms the resurrection of Woolly Mammoths is possible. Shapiro says that there are two phases in de-extinction: “The first phase includes everything up to the birth of a living organism, and the second will involve the production, rearing, release, and, ultimately, management of populations in the wild.”
These 12 steps are the first phase in the Woolly Mammoth de-extinction process:
Step 1: Collect Asian Elephant DNA
The first step is DNA extraction using a blood or tissue sample from a living animal. DNA extraction is a well-established method to isolate DNA from a cell’s nucleus. The DNA is purified to extract debris and then tested for quantity and quality.
Step 2: Sequence the Asian Elephant Genome
Sequencing DNA involves establishing the order of a DNA molecule structure’s four bases or chemical building blocks. For example, DNA sequences show which sections contain genes and which sequences hold the regulatory instructions that turn genes on or off. The Colossal Team and Dr. Church have already sequenced 23 Asian Elephant genomes.
Step 3: Collect Viable Woolly Mammoth Tissue Samples
The last Woolly Mammoths died a millennia ago, but there are highly well-preserved remains due to their icy habitat. Those remains are suitable for DNA extraction, although some of the sequences may be fragments because of their age. In 2018, Dr. Church and his colleague Dr. Eriona Hysolli acquired significant Woolly Mammoth tissue samples in Chersky-Siberia.
Step 4: Sequence the Woolly Mammoth Genome
Dr. Church and his colleague identify the genetic difference between Woolly Mammoths and modern Asian Elephants. With both types of samples, the Colossal teams compared the two genomes and inserted critical factors of the Woolly Mammoth genome into the Asian Elephant genome. They have sequenced the Woolly Mammoth genome.
Step 5: Identify Traits to Edit within the Asian Elephant Genome
They collect traits that will enhance the ability to resist the cold. Those cold-resistant traits include smaller ears, shaggy fur, hemoglobin adapted to cold, and excess fat tissue. These are the Woolly Mammoth traits that will be inserted into the Asian Elephant genome.
Step 6: Build CRISPR Libraries that Will Allow for Editing Identified Genes within the Asian Elephant Genome
CRISPR libraries contain synthetic Woolly Mammoth DNA. Before inserting the Woolly Mammoth DNA into the Asian Elephant genome, a CRISPR library holds the Woolly Mammoth DNA and encodes over 50 identified cold-resistant traits.
Step 7: Insertion of CRISPR Libraries
Science teams insert the modified cold-resistant genes into the genome of an Asian elephant. Once in the genome, a CRISPR/Cas9 complex containing RNA binds to the identified gene in the Asian Elephant genome, cutting into the double strands of the Asian Elephant DNA. This step allows for the hybrid DNA insertion in its place. This cut and paste process repeats for each gene. Each new cell replication then expresses the hybrid gene to pass on to future generations. The hybrid gene is an Asian Elephant gene-edited with Woolly Mammoth DNA to enhance the desired cold-resistant traits.
Step 8: Verify that the Cold Resistant Traits Are Expressed in Hybrid Cells
The hybrid genes need to be tested and verified. Cold-resistant trait testing involves the use of various assays. Assays are qualitative and quantitative analyses to determine the presence, amount, or functional activity of a target gene or trait.
Step 9: Embryo Transfer
This step uses SCNT. Once Colossal’s scientists are satisfied that the hybrid cells are functioning correctly, the nucleus transfer begins. The nucleus from a donated Asian elephant egg is removed, and the hybrid nucleus, which is the Asian elephant nucleus edited with the woolly mammoth DNA, is inserted in its place. Electrical pulses are applied to the egg to stimulate fertilization. The egg then begins to divide and grow into an embryo.
Step 10: Implantation
The modified egg, containing the woolly mammoth DNA, is further developed outside the body while it grows into an embryo. After the embryo has developed, it is implanted into a surrogate African elephant, where it will be carried to term.
Step 11: Gestation
Gestation, or growth and development in the womb, usually takes 18 – 22 months in African elephant species.
Step 12: The Culmination is the Birth of the Woolly Mammoth
The newborn will be a hybrid with genetic traits from the extinct Woolly Mammoth and the Asian Elephant, its living relative.
Science and Colossal are close to the de-extinction of the Woolly Mammoth. Many of the steps are complete or in the process of refinement. It is critical to have a solid foundation in saving, conserving, and ultimately making species that do more than marginally survive. We are building the bridge that eventually crosses that divide to a better, healthier world.
The Woolly Mammoth Is Only the Beginning of Colossal De-Extinction
At Colossal, we’re building a DNA database, so no living creature faces the theft of their existence. Through technological breakthroughs, we’ll advance genomics. Today, the Woolly Mammoth. Tomorrow, we may hold cures for blindness, tumor, and disease eradication. Many companies claim they’re going to change the world. At Colossal, we believe that the world doesn’t need to be changed. The world needs to be healed.
Interested in Following the Woolly Mammoth De-Extinction?
Woolly Mammoth de-extinction spurs advances in genetic engineering and will also be a force to undo hundreds of years of ecological damage that the human species has inflicted on planet earth.
Let’s make a better world together. Join our newsletter for updates.